
Thus, Cl has an oxidation state of +5 in this compound.Īlso, this is a website that could help you to check your work. In the case of KClO 3: KClO 3 and KCl are both ionic compounds with the cation K+, while O has its typical oxidation number of −2. Because KCl is a neutral ionic compound, the sum of the oxidation numbers must be 0. Because Cl can have more than one charged state, we determine its charge, and therefore its oxidation state, based on K. Hence the ionic charge of Chlorine (Cl) is 1. There is a nice series of halogen oxyanions.and as always the oxidation number is the charge left on the central atom when all the bonding pairs of electrons are broken with the charge assigned to the most electronegative atom. 
And as the Chlorine (Cl) gains 1 electron, it forms Cl ion. In 'potassium chlorate' we gots Cl(+V).and O(-II).and also K(+I). Since there are an equal number of atoms of each element on both sides, the equation is balanced. 
During the chemical reaction, chlorine gains 1 more electron and achieves the nearest noble gas configuration to become stable. Step 3: Verify that the equation is balanced. Place the ions in their proper order: cation and then anion. The procedure for naming such compounds is outlined in Figure 2.10 'Naming an Ionic Compound' and uses the following steps: Figure 2.10 Naming an Ionic Compound. K belongs to Group 1 on the periodic table so would lose an electron in order to reach a full valence shell. You can see that the outermost orbit of Chlorine has 7 electrons. We begin with binary ionic compounds, which contain only two elements. 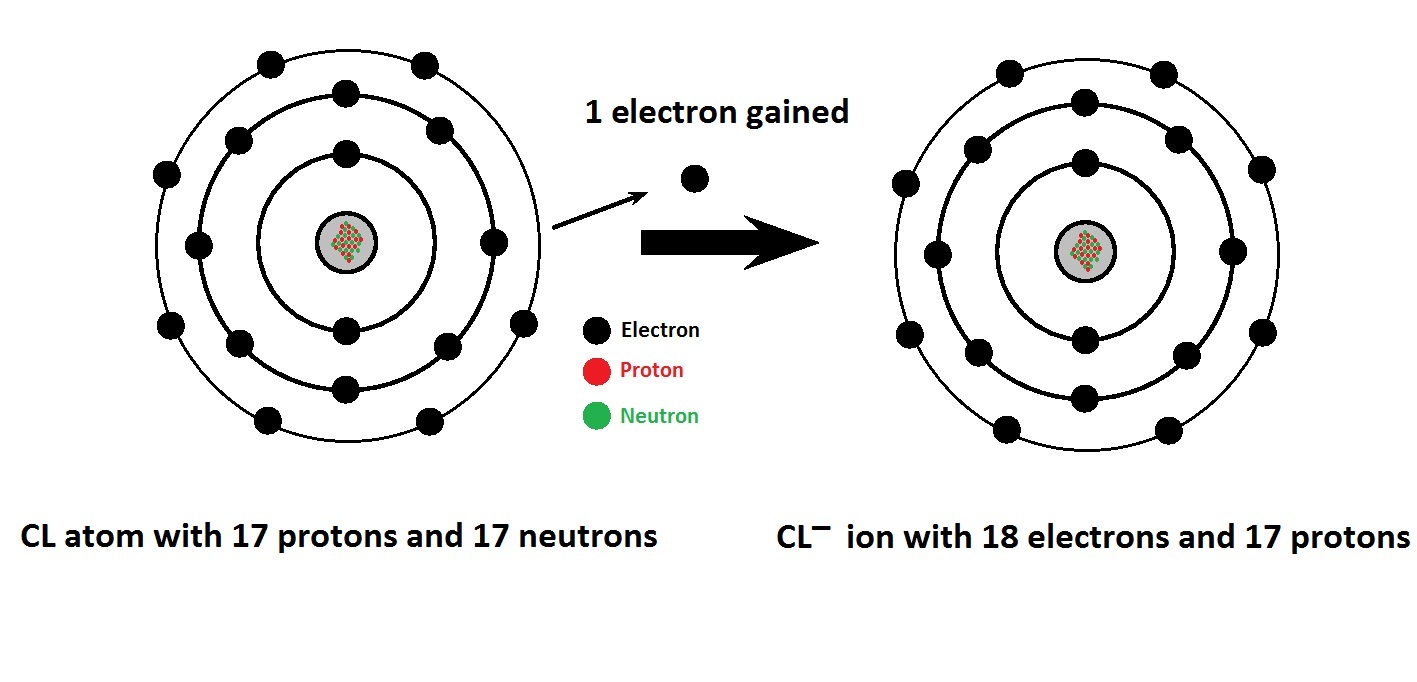
So in the case of KCl: We look at K first because it has one charged state. To find the correct oxidation state of Cl in KClO4 (Potassium perchlorate), and each element in the molecule, we use a few rules and some simple math.First. To predict the stable charge of a non-transition metal that has only one charged state, you remove enough electrons so that the cation or anion has the same number of outer shell electrons as a noble gas, that is, it is isoelectronic to the noble gas. It is helpful to remember that monoatomic ions always have an oxidation number equal to their charge. Though through frequent and repetitive practice, it'll probably happen anyway. You can definitely just have the Periodic Table by your side so that you don't really have to memorize oxidation numbers. it gained electrons).Arisawaters2D wrote:Is the best way to go about these types of problems memorizing the oxidation numbers for some elements? it lost electrons) and KClO 3 is a oxidizing agent (i.e. For potassium hypochlorite (KClO), oxidation state for each atom is. oxidation number goes down).ĢKClO3 = 2KCl + 3O2 is a redox reaction where O is oxidized and Cl is reduced. What is the oxidizing agent? The oxidizing agent, or oxidant/oxidizer, is a species that oxidizes other substances and gains electrons (i.e. The higher oxides also react as a mediator in HCOOH oxidation in competition with O2 evolvement. Count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges/ions) are balanced. Then IrO3(Cl) or RuO3(Cl) reacts with Cl-to produce Cl2 and regenerate the adsorbed hydroxyl radicals. What is the reducing agent? The reducing agent, or reductant, is a species that undergoes oxidation loses or "donates" electrons (i.e. Substitute Coefficients and Verify Result. Oxidation is the loss of electrons or an increase in the oxidation state of an atom, ion, or certain atoms in a molecule. Reduction is the gain of electrons or a reduction in the oxidation state of an atom, ion, or certain atoms in a molecule. Next, find the number of each atom that changed:įind the difference in electrons using the product of the above tables: Find all the pairs of atoms of the same element where the oxidation state has changed:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
